Urgent Antibiotic-Resistant Threats: Carbapenem resistant Enterobacteriaceae (CRE)
Introduction
Most of us have heard how over the last 60-80 years antibiotics, by removing the threat of many infectious diseases, wiped out a main cause of child mortality and improved and prolonged our lives. However, in recent years many of us have also heard alarming stories that some of these diseases may become un-treatable again because of increasing resistance to antibiotics. Will the so-called post-antibiotic era arrive soon? Is it possible for the days of nightmare to return when effective antibiotics are no longer available to keep us out of harm’s way? In some instances, this is already happening, for example in the case of Carbapenem-resistant Enterobacteriaceae, a group of bacteria.
The Centers of Disease Control (CDC) categorizes the top 18 threats posed by drug-resistant microorganisms into three classes: urgent threats, serious threats, and concerning threats. There are only three examples of urgent threats: Clostridium difficile, carbapenem-resistant Enterobacteriaceae (mainly Klebsiella pneumoniae and E .coli), and Neisseria gonorrhoeae. The best-known hazardous organism, methicillin-resistant Staphylococcus aureus (MRSA), is now classified as a serious but not urgent threat. Carbapenem-resistant Enterobacteriaceae (CRE) have become resistant to nearly all the antibiotics we have today. Almost half of hospital patients who get bloodstream infections from CRE bacteria die from the infection.
The following is a short report on CRE.
Imipenem, a member of the carbapenem class of antibiotics
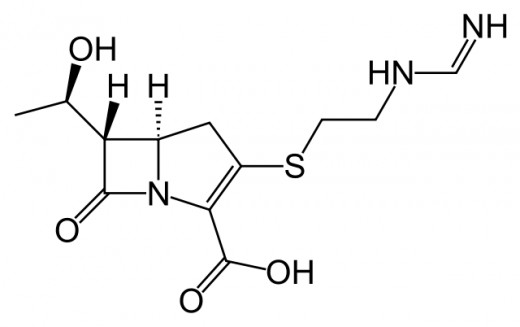
What is Carbapenem?
Carbapenems, like penicillin, contain a chemical structure called the beta-lactam ring that binds to, and kills an enzyme involved in bacterial cell wall synthesis. A bacterium cannot survive without an intact cell wall.
The first beta-lactam ring containing antibiotic was penicillin. Bacteria that are resistant to penicillin and its derivatives have been known for a long time. With the widespread use of beta-lactam antibiotics, more and more organisms became resistant to them, and this necessitated the introduction of newer and newer antibiotics. Some of these also work like penicillin, but subtle differences between the structures of the drugs make them more or less sensitive to the effects of the bacterium’s defense arsenal. Carbapenems have a structure that renders them resistant to breakdown by enzymes that attack most beta-lactam rings.
Thus, carbapenems were considered the antibiotics of last resort of many infections, such as those by Escherichia coli (E. coli) and Klebsiella pneumoniae. Unfortunately, as the title of this article indicates, many strains have developed resistance to carbapenems.
What are Enterobacteriaceae?
Enterobacteriaceae is a large family of bacteria that includes many harmless organisms, as well as several pathogens. E. coli and Klebsiella pneumoniae are the two main species belonging to this family that cause widespread problems. Both are normally present in the human gut, but if they colonize other organs they can cause serious infections.
Klebsiella pneumoniae
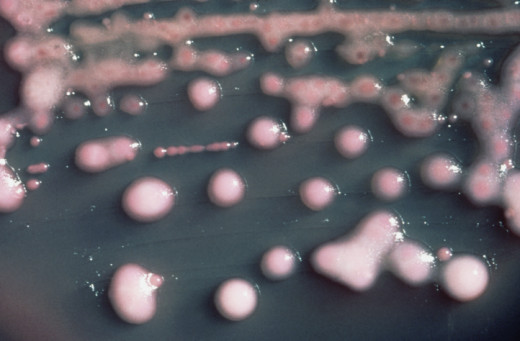
What diseases does Klebsiella pneumoniae (K. pneumoniae) cause?
K. pneumoniae has been recognized for long as a risk factor for pneumonia, especially in immunocompromised individuals. It can also cause urinary and biliary tract infections, as well as wound site infections. The range of diseases caused by K. pneumoniae is wide, with a number of conditions being serious or life-threatening, such as meningitis and septicemia.
Escherichia coli - Scanning Electron Microscopy

Which diseases does E. coli cause?
Most strains of E. coli are harmless, but some cause food poisoning. The harmless strains are part of the normal bacterial components of the gut. Although there is a multitude of other organisms that are risk factors for urinary tract infections, E. coli is the most common cause of complicated infections (so called uropathogenic E. coli). Some E. coli strains are known to produce a toxin (Shiga toxin or verotoxin) that destroys red blood cells, and causes a hemolytic-uremic syndrome (destruction of red blood cells and kidney failure). In May 2011, there was an outbreak in Germany, caused by a Shiga toxin- producing strain. In addition, E. coli can cause meningitis in newborns.
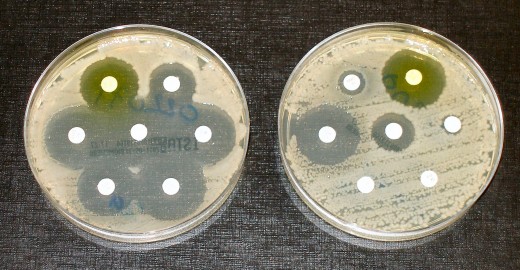
What are the mechanisms by which organisms become resistant to carbapenem?
What are the mechanisms by which organisms become resistant to carbapenem antibiotics?
Bacteria can become resistant to carbapenem (and to antibiotics in general) by a number of mechanisms:
----◊ By acquiring the ability to transport the antibiotic out of the cell
----◊ By changing the outer membrane structure in a way that the antibiotic cannot penetrate the cell
----◊ By gaining enzymes that break down carbapenems. Several classes of such enzymes exist. These arise in the following ways:
--- Through mutations in pre-existing enzymes
--- By increased production of existing enzymes that inactivate the antibiotic
--- By uptake of DNA that codes for enzymes that inactivate the antibiotic. This DNA is usually located on mobile elements (pieces of DNA such as plasmids that are able to propagate themselves and move from one bacterial cell to another). Mobile elements represent the fastest way for antibiotic resistance to spread among bacteria. Plasmids can sometimes carry multiple genes each coding for an activity that can disarm a different class of antibiotic.
It seems there is an amazing capacity for bacteria to find many different ways to disable or expel antibiotics! However, we have to remember that antibiotics are naturally occurring compounds that are environmental toxins for bacteria, and bacteria have been able to develop strategies to combat them over millions of years of evolution. When a new antibiotic is at first introduced in medicine, it kills most of the bacteria, since they are sensitive to it. Unfortunately, a few of the bacteria are not killed because they are resistant to the antibiotic. These can grow and multiply until they largely replace the sensitive bacteria.
An outbreak of food-borne E. coli infection in 2011 was most serious in Germany, causing 54 deaths.
What are the major risk factors?
These infections mainly affect people in hospital or long-term care settings. Patients in the greatest danger of contracting CRE are those with weakened immune system, or people who use invasive devices (catheters, or are on mechanical ventilation etc). Cases in which the infection spread by reusing of duodenoscopes (an endoscope for examining the duodenum) have been reported.
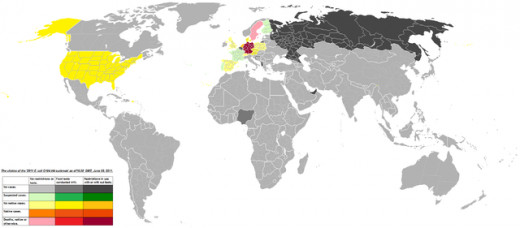
The food industry can be the source of drug-resistant organisms. The cause of the German outbreak in 2011 (see above) was, in all probability, fenugreek seeds imported from Egypt. This outbreak affected almost 4000 people, with 845 developing the serious hemolytic uremic syndrome, and 54 dying. The administrative management of this outbreak was recently analyzed in Nature magazine. Among the improvements that could have ameliorated the situation were a more flexible reporting system, better coordination amongst agencies and more efficient methods for the identification of bacterial strains; see http://evahard.hubpages.com/hub/Antibiotic-Resistance-The-Presidential-Order, on directives to develop fast detection methods in this country. As the map shows, the outbreak mainly affected Germany. Note that the original title of the map is “Toxic cucumbers” which is incorrect. The map was posted on June 7, 2011, whereas the real origin of the E. coli strain – fenugreek seeds – was not announced publicly until July 5. The outbreak started on May 19. Also note that the strain responsible for the outbreak was resistant to many antibiotics but not carbapenem. Antibiotic treatment was not highly recommended for treatment, because it increased toxin production by the bacterium.
Animals are also considered to be a source of antibiotic resistant organisms, both through handling and through consumption.
Treatment
Some newer antibiotics, for example phosphomycin and tigecycline, can treat diseases caused by multiresistant bacteria. Not all patients respond well to treatment, as is evident from the high mortality rate. This is not only because of the poor health of patients in long-term care. A given antibiotic may not work efficiently enough for all varieties of the disease. For example, tigecycline is not an efficient therapy for bloodstream infections even though CRE organisms are sensitive to it, because after intravenous injection the antibiotic quickly diffuses into the tissues, resulting in too low a plasma concentration.
Prevention
Protocols for prevention, including the isolation of infected patients, updated protocols for cleaning surfaces etc. are in place in hospitals.
Careful hand washing and washing of uncooked vegetables and meat are crucial to fend off infection. Food poisoning is possible through these sources. Thorough cooking of meat is also important, especially meat that cannot be washed such as hamburger meat (the latter is this author’s personal opinion).
If you have procedures necessitating the use of a duodenoscope, for example endoscopic retrograde cholangiopancreatography (ERCP), talk to the healthcare professionals about concerns.
Update
In a recent (March 2016) issue of Science, the “Insights” section presented further warnings on the spread of extremely antibiotic resistant CRE. Colistin, an antibiotic that was developed about half a century ago but was not widely used because of its toxicity, has recently been reemployed as an emergency measure to fight multiresistant CRE organisms. However, organisms resistant to colistin itself have already appeared. The “Insights” review refers to recent studies showing that colistin resistance, the so-called mcr-1 gene, can be carried on plasmids, which is not good news because this mechanism greatly facilitates its spread.
Sprenger M., Fukuda K.: New mechanisms, new worries. Science 2016; 351: 1263-1264
Sources
http://www.cdc.gov/drugresistance/biggest_threats.html
Witze A, Morello L, Turner M. Scientific advice: Crisis counselors. Volcanic eruptions, oil spills and bacterial outbreaks all land in the laps of government science advisers, and put them to the test. Nature 2014; 512: 360-363
Wikipedia: Carbapenem-resistant Enterobacteriaceae
Blair JM, Webber MA, Baylay AJ, Ogbolu DO, Piddock LJ. Molecular mechanisms of antibiotic resistance. Nat Rev Microbiol. 2015; 13: 42-51.