Cancer Stem Cells- The New Frontier
A lot of my article topics come from previous work experience. I’ve written a few articles on cancer based on my experience in a drug research lab where the target was often a subset of cancer. Another topic I am always fascinated with, again due to a brief time working with the agent in question, is stem cells. So imagine my surprise towards the end of last year when I began to see casual references to the idea of cancer stem cells in my current work. I was immediately intrigued and put it on my list of things to look into.
A common theme in research is that the idea often seriously predates the proof; cancer stem cells follow this trend. Although hypothesized as far back as 150 years ago, with momentum gaining in the 60s, definitive proof of cancer stem cells didn’t come until 1997. Cancer itself is a very broad term with each subdivision named for the tissue or organ where the cancer originates. While the possibility presents itself that each cancer stem cell responds to the same types of microenvironment signaling, the distinction is made for each different type of cancer having its own stem cell. To dive into the specifics let’s look at two different types of cancer, Breast Cancer and Leukemia, and see how their stem cell research is coming along.
To back track a little let’s be clear on what stem cells do. These cells have two key functions that make them unique (but not special). Stem cells are not specialized cells, what makes them unique is that they don’t have a specific function in a given tissue instead they excel at self-renewal and giving rise to cells that will ultimately become specialized. Upon division stem cells have two options- the first is the daughter cell can become a stem cell, a carbon copy of the parent cell. This option allows stem cells to extensively self-renew as they can proliferate over and over to repopulate. The second option for the daughter cell upon division is to become a more differentiated cell. Stems cells represent the far end of the spectrum in terms of specialization and can differentiate into more specialized cells until they reach the opposite side of the spectrum, very specialized without the ability to self-renewal. Differentiation is the act of taking an unspecialized cell and making it more specialized. This is generally a multistep process starting with a stem cell, going to a progenitor cell-a more targeted but still multipotent cell- and so on until the most specialized cell is created, for example a red blood cell.
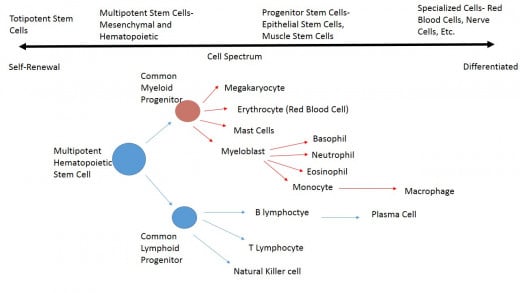
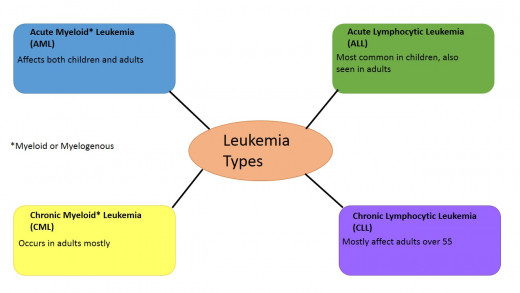
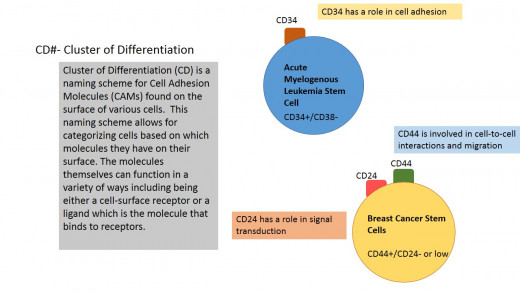
The cancer phenotype is not only different depending on the origin tissue or organ, making Leukemia an entirely different disease than Breast Cancer, but also within the given type of cancer there are often distinct phenotypes. In the case of Leukemia the first question is whether the Leukemia is acute or chronic, next is which progenitor blood cells did the cancer originated in. In the case of Lymphocytic Leukemia the origin cells are lymphocytes while the myelogenous (or myeloid) type is derived in myeloid cells which includes red blood cells, platelets, and the granulocytes. Leukemia stem cells (LSCs) were the first cancer stem cells to be discovered proving the theory hundreds of years after the idea was first speculated on. Specifically it was the Acute Myeloid Leukemia (AML) stem cells that were discovered at the University of Toronto by researchers Bonnet and Dick. They isolated cells from (human) tumors and implanted them into immunodeficient mice. The cells capable of initiating tumors in the mice were then phenotypically found to be CD34+/CD38- in regards to the cell's surface. These cells possess the major stem cell characteristics- the ability to self-renew via proliferation and the ability to give rise to more specialized cells via differentiation. These CD# molecules are cell-surface markers with various functions. The naming system of referring to them as CD# is a way of identifying specific cell types, mostly stem cells and blood cells. CD34 is the major marker found on hematopoietic stem cells, the stem cells that give rise to all blood cells. CD34 is a membrane protein that functions in which probably aids stem cells in binding to various locations within the body such as the bone marrow. CD38 is largely expressed on leukocytes, it has a role in cell adhesion and signal transduction including calcium signaling.
Since the discovery of the AML stem cells there have been discoveries of additional stem cells for other types of cancers including Breast Cancer, Brain Cancer, and Pancreatic Cancer. A major distinction between cancer types is whether they are blood cancers or solid tumor cancers. Once a blood cancer was found to have cancer stem cells it spawned the question whether solid tumor cancers also possess cancer stem cells. Al-Hajj et al. applied Bonnet and Dick’s method of harvesting Breast Cancer patient’s cells to determine which subpopulations could initiate cancer when transplanted into immunocompromised mice, then they determined the common characteristics of the cells that did result in tumors. This proved the theory that along with blood-based cancers, solid tumors also possess their own cancer stem cells.
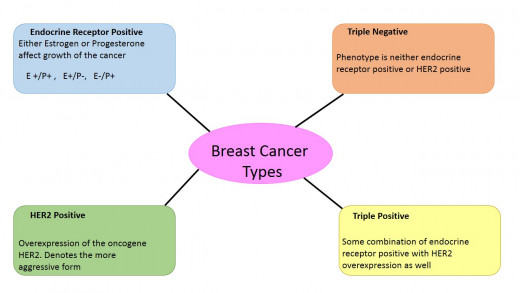
Similar in classification to Leukemia, Breast Cancer is divided into several categories – Endocrine Receptor Positive (either Estrogen, Progesterone, one, or both); HER2 positive (overexpression of Human Epidermal Growth Factor Receptor 2); Triple Positive (Estrogen, Progesterone receptor positive along with overexpression of HER+); Triple Negative (none of the above). These phenotypes respond differently to various treatments and thus are generally treated as separate subclasses of Breast Cancer. The Breast Cancer Stem Cell, generally denoted as B-CSC, has several defining characteristics. Similar to the LSC it has two cell surface markers to note- CD44+/CD24-/ low (where negative/low indicates the cells either have very little or no CD24 on their surface). CD44 is cell surface glycoprotein involved in cell adhesion, cell-to-cell interactions, and migration; while CD24 is also a glycoprotein involved in signal transduction.
After the initial discovery of these two markers with the AML experiment adaption, additional markers for Breast Cancer Stem Cells were identified. Research indicates Breast Cancer Stem Cells have also been found to overexpress aldehyde dehydrogenase (ALDH). These characteristics don’t always overlap, some B-CSCs have been found to either overexpress ALDH or have the CD44+/CD24-/ low phenotype for example. ALDH has been found to metabolize chemotherapeutic agents, such as cyclophosphamide. Current research indicates the Triple Negative form of Breast Cancer is the phenotype that is most stem-like. Research has been done to confirm B-CSCs resistance against the major cancer arsenal- chemotherapy, radiotherapy, and hormone therapy. This last point illustrates why this discovery and continued research into this class of stem cells is so important.
The discovery of these cancer-specific stem cells along with increased understanding of these cancer cell populations could have limitless potential in the diagnosis, treatment, and prevention of the type of cancer itself. Cancer relapse and treatment failure are hypothesized to be the work of these cancer stem cells. The way to illustrate this point is to think of a tumor as having two distinct populations of cells, cancer stem cells and ‘regular’ cancer cells. Researchers have known about these regular cancer cells for years and thus the cancer drugs out there are targeting these cells. It should come as no surprise that many cancer drugs, like chemotherapy agents, target the cell cycle since cancer is synonymous with overgrowth. One key characteristic of stem cells is they are largely quiescent, that is to say they don’t divide on a regular schedule and thus these CSCs largely avoid detection by, and destruction via, chemotherapeutic drugs.
Not only are these CSCs resistant to treatment but they are most likely the culprit in why cancer comes back since they are left behind in the body after treatment has ceased. After cancer treatment successfully wipes out the normal cancer cells, these remaining cancer stem cells now have plenty of room and resources to divide, differentiate, and present the body with a new tumor. Now that researchers have caught onto this charade they are hard at work looking into drugs, both new and old, that will knock out these stem cells. A combination of traditional cancer medicine (to attack the ‘regular’ cancer cells) plus another agent or two that specifically targets these troublemaker CSCs should have a much better effect on the cancer phenotype including remission. Cancer stem cells are also the suspected culprit in tumor metastasis since these cells have displayed highly invasive properties, this is also currently being investigated and could provide additional therapeutic angles for metastatic cancer.
Resources available on request.