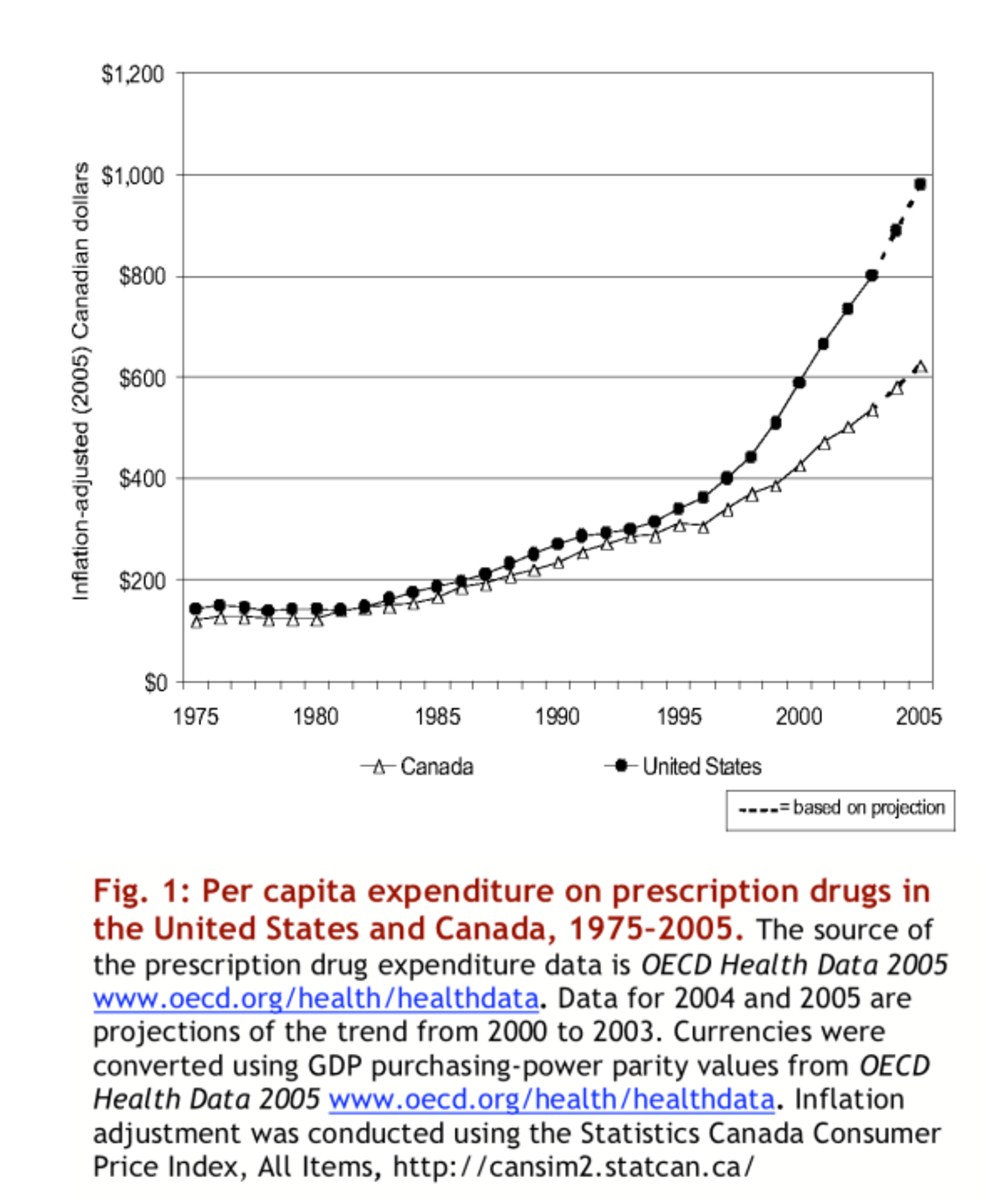
Off-Market Label Drugs Deceive Patients
If Life Could be so Simple

Ill-Health Prevention Wellness Programs Include Good Pain Mgt & Fit Techniques
If It's a Man Made Drug, Their is Health Risk
I’ve stated before, “Anything manmade and outside of God’s natural organic order, can/will cause ill-health medical conditions.” And there is nothing more true to this statement and applicable relative to the plethora of FDA approved drugs that are causing great harm to those with medical conditions and dependent on pharmaceuticals.
I believe there is no manmade drug (including long-storage processed/restaurant, fast-foods) that is entirely good for us, I would concede there are medical conditions where specific types of drugs given to patients far outweigh the health risk consequences than going without. My biggest concern with our FDA (Federal Drug Administration) approved drugs process is the sheer number of them coming to market.
Recall, before these drugs come to market there is an extensive FDA safety-approval process that also equates to billions of R&D (Research & Development) dollars where investors demand a ROI (Return on Investment). And it is also true that that much of the Federal Drug Administration budgets are greatly dependent upon the pharmaceutical products and services industries. These industry resources also equate to 10’s of thousands of government and special interest jobs, services and programs within our health care industry.
Now part of the medical class action lawsuits incorporate the cost-of-doing business with drugs that have caused severe health conditions and early death. The pharmaceutical companies are now using Off-Market Labels that don't require FDA approval to continue selling the same dangerous drugs for different conditions. (Read related linked hub below, "Humans, Drug Lab Rats? And also click link to hub below and read about the 41 class action lawsuits).
It is my opinion, the drug companies know full well how to fudge research data and the FDA knows how to give a free pass. Folks this is a dangerous combination for the unknowing patient in need of safe effective drugs with minimal side effects. We already know anything manmade is going to create additional risk for our health. However, how much unnecessary risk should the “unknowing” patient take? Are drugs that create high risk health conditions with “little-to-no” health benefits really necessary? And has our drug approval process “now” incorporated increased health risk conditions and acceptable death rate into the cost-of-doing business? And is litigation and class action lawsuits now factored into the cost of doing business as well?
It would appear, “patients” have become a part of the “rat” lab program included into the cost or R&D “in the open market!” Human studies also seem to be a legitimate process that incorporates reuse of the same drug under different brand names (read below “Off-Market Labels ”) to treat conditions that were not initially approved by the FDA. I submit… The cost of doing business in expediting drug-to-market approval for use on humans is more cost effective with better correlated data than animal lab studies.
It seems the paradigm shift is Humans are an expendable commodity where the cost of doing business is settled through class action lawsuits to appease suffering families by compensating financially. Also more research animals are saved, simultaneously keeping activist at bay; investors also make huge fortunes; government and private sector jobs and special interests continue to thrive and grow.
Have we become the little white rats supporting the Off-Market Label drug trials as drug products are expedited for use in the marketplace? And is this why our prescription drugs have a ridiculously high cost: “Factor cost of class lawsuits and off-label product marketing costs and FDA blind-eye policy kickbacks to give a green light on drug re-distribution .” Are Off-Market Label drug policies breaking our healthcare systems back? Is there anything that can be done about it?
Look for answers in the related hub link I've provided below "Humans, Drug Lab Rats ?.
- Humans, Drug Lab Rats?
Have we become the little white rats supporting the Off-Market Label drug trials and product-to-market programs? And is this why our prescription drugs have a ridiculous cost: Factor cost of class...