Pharmacodynamics and Pharmacokinetics Based Dosing of Antibiotics in Medical Literature
There has been an increasing interest in Pharmacodynamics and Pharmacokinetics (PK/PD) based dosing of antibiotics in medical literature. Nevertheless, once daily dosing of Aminoglycosides is perhaps the only translational change based on PD based dosing in clinical practice. Despite the availability of a wealth of literature on PK/PD based dosing, limited data is available to support a direct relationship of this approach with positive clinical outcomes. Nevertheless, pharmacists working or studying Infectious Diseases Pharmacotherapy should have a meaningful understanding of this current topic.
It is increasingly important in developing new strategies to retain the effective use of existing antimicrobials to address the increased resistance with both gram positive and negative bacteria because there are no new antibiotics in the pipeline. [1] Current Antimicrobial Stewardship guidelines recommend pharmacodynamic-based antibiotic dosage optimization strategies to be a great approach in improving the effective utilisation of existing antimicrobials. [1] Identifying the relationship of antibiotic concentration to microbial potency and its antibacterial effectiveness and activity helps develop the optimal dosing regimes needed to effectively kill bacteria in clinical practice. [1] Minimising inappropriate use and improving antimicrobial selection, route, dose and duration of therapy helps reduce resistance and promotes optimized use of antimicrobials for optimal clinical outcomes.[1]
The laboratory investigation of antimicrobial activity is vital in the pharmacotherapeutic management of infectious diseases. [1]Clinicians’ understanding of basic pharmacodynamic principles and laboratory tests like minimum inhibitory and minimum bactericidal concentrations tests, time-kill tests, post-antibiotic effects tests and antimicrobial combination testing are important in helping determine antimicrobial pharmacodynamic properties and promote optimal effective treatment.[2]
For example, a fixed drug dose administered to a big population will result in significantly different profiles to when drug concentrations are altered over time. [1] Different drugs display different pharmacodynamic properties and only free non-protein bound drug is microbiologically active. [1]Higher values of the measure of drug potency (such as the minimum inhibitory concentration or MIC) for the pathogen indicate that a fixed drug exposure will have lesser effect [1] Antimicrobial effect is dependent upon drug exposure at the infection site. [1] The bactericidal activity of antimicrobials can be differentiated through the concept of time and concentration relative to drug exposure.[1] Some relationship parameters to determine antimicrobial activity include the mathematical relationship between antibacterial effect and the maximum free drug concentration/MIC ratio (Cmax/MIC) , the area under the free drug concentration-time curve to MIC ratio (AUC/MIC) or the time in which free drug concentrations exceed MIC(∫T>MIC) with microbiological outcome. [1] AUC refers to the actual body exposure to the drug after administration. [1]
Two unique examples that clearly demonstrate relevance of the gained knowledge to pharmacy practice (Practice Implications and relevance to reflect on my practice):
Different antibiotics work optimally through varying dosing regimens, therapy duration, drugconcentrations etcetera. [1] Currently, there are definedoptimal dosing strategies for many available antibiotics used frequently such as beta-lactams and aminoglycosides which are useful in promoting optimal therapeutic outcomes. [1] Pharmacodynamic profiles of available antimicrobials are either concentration-dependent or time-dependent. [1]
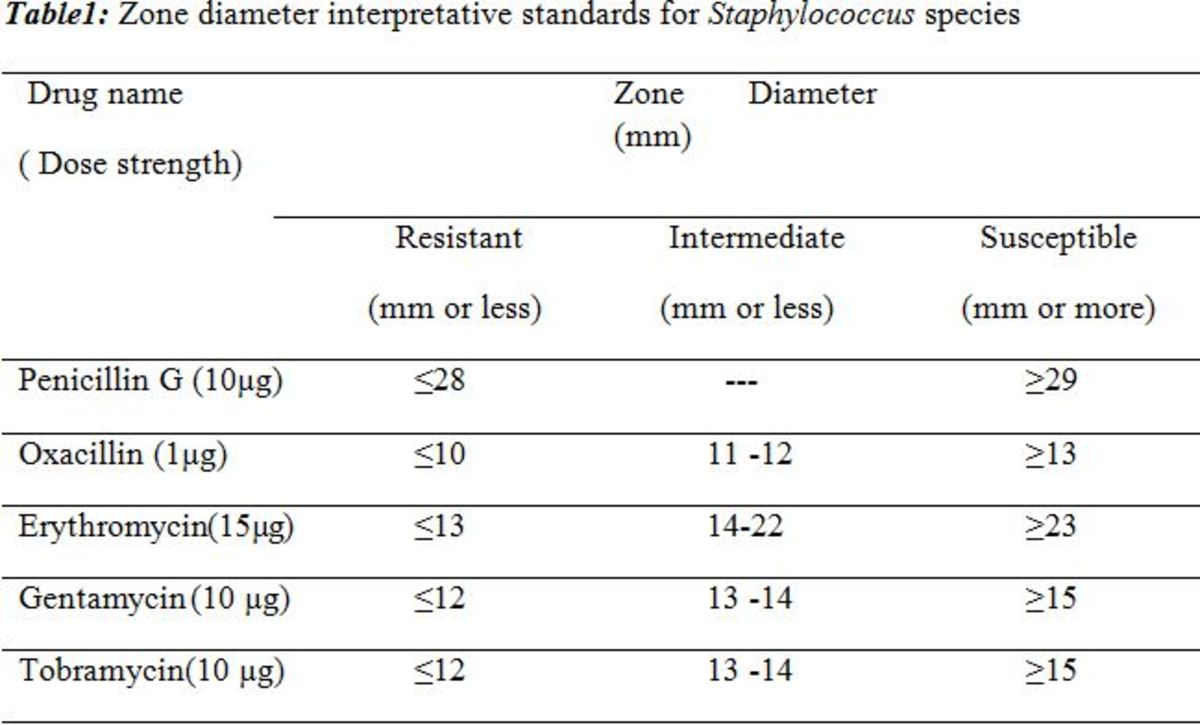
For example, aminoglycosides activity depends on maximising peak free drug concentration in relation to MIC thus higher doses given less frequently is the gold standard strategy for optimizing their pharmacodynamics. [1] Monitoring serum aminoglycoside concentration and using extended-interval doses helps maximise efficacy and minimise aminoglycoside-related toxicity for certain infections through preventing drug accumulation. [2] A nomogram approach involves the example of “using a fixed 7mg/kg intravenous gentamicin dose with a dosing interval nomogram based on patients’ estimated creatinine clearance (Hartford nomogram)” which observed similar clinical response rates to historical data but with a reduced nephrotoxicity. [1] Doses used in different hospitals using a once daily aminoglycoside strategy are individualized based on gentamicin or tobramycin MICs against P.aeruginosa. [1] The MIC of the pathogen is the major factor in determining the approach to which optimal drug and dose regime is utilised because based on the mathematical ratios and equations, one-dilution change in MIC results in doubling or having the pharmacodynamic exposure. [1] For pathogens with lower MICs of below 2mcg/mL, the nomogram approach is better for resulting in adequate required therapeutic exposure whereas greater MICs and variability in pharmacokinetic profiles of patients (e.g. renal function) would benefit from a more individualized treatment approach using therapeutic drug monitoring and MIC. [1]
Thus once-daily aminoglycoside dosing is both effective and a well-established approach to enhance therapeutic efficacy while decreasing toxicity risk and improving compliance through simplified dosing and monitoring. [3]
The pharmacokinetic and pharmacodynamic profiles of drugs change as people grow up and this can markedly change drug effects. [4] A prospective study investigating how PK/PD modelling of dosing amikacin, morphine and busulfan in children resulted in optimization and individualization in paediatric clinical care and revealed insights into organ system maturation. [4] This approach leads to predictable safety and efficacy of medicines in all ages. [4]
Soas to prescribe medicines guided by an evidence-based method, a thorough understanding of pharmacological profile of patients are required since there is a large variation of drug response between adults and children as well as in those with co-existing conditions like renal or hepatic impairment for example. [4] Moreover, understanding the pharmacodynamic effects of drugs on our bodies is imperative for evidence-based medicine and optimal therapeutic effects and safety. [4]
The second example of the application of optimizing therapy based on pharmacodynamics and pharmacokinetics include population-PK modelling that resulted in individualised dosing regimen for antibiotic amikacin that is largely renally eliminated through glomerular filtration in a large population of more than 800 preterm and term neonates.[4] Pharmacokinetic data revealed that in contrast to current paediatric dosing regimens, older children with higher weight had a faster maturation of clearance and proposed that the evidence-based dosing regimen be guided by birth weight combined with postnatal age to decide on appropriate dose rather than rely on current weight. [4] They compared the simulated concentrations of the current paediatric regimens versus the new dosing regimen and reported that the new evidence-based model was superior in achievement of target peak and trough concentrations and this model-based individualised dosing regimen is expected to prevent medicine-associated toxicity while maintaining efficacy. [4]
Reflection
Through the knowledge I have gained in this task, I have a better understanding of how different antimicrobials exert their therapeutic activity and how individualised dosing based on pharmacodynamics and pharmacokinetics and taking into account individual patient characteristics is an effective evidence-based medicine tool to promote optimal efficacy and safety of using medicines in practice. I also have a better understanding of how the MICs of drugs with respect to offending pathogens can better guide therapeutic regimens for all patients and guide hospital therapeutic regimens and therapeutic dosing protocols to minimise antibiotic resistance and promote the predictable safe and effective use of existing antimicrobials.
REFERENCES
1) Connors KP, Kuti JL and NicolauDP.Optimizing antibiotic pharmacodynamics for clinical practice.Pharmaceut Anal Acta.2013;4(3):1-8
2) DiPiro J, Talbert RL, Yee GC, Matzke GR, Wells BG and Posey LM. Pharmacotherapy-A pathophysiologic approach 10th edition. United States of America. McGraw-Hill Education;2017.Section 16
3)StankowiczMS, Ibrahim J and Brown D.Once-daily aminoglycoside dosing:an update on current literature.AmJHealthSystPharm.[Internet].2015 Aug;72(16):1357-1364. Available from: http://www.ajhp.org/content/72/16/1357.long?sso-checked=true
4) Admiraal R, Van-Kesteren C, Boelens JJ, Bredius RG, Tibboel D, Knibbe CA. Towards evidence-based dosing regimens in children on the basis of population pharmacokinetic pharmacodynamic modelling.Arch Dis Child.[Internet]. 2014 Mar;99(3):267-72. Available from: http://adc.bmj.com.ezproxy.utas.edu.au/content/99/3/267.long