The Basics of Clinical Trials

The Drug Discovery and Development process is a long, complicated, and interesting path. The average time it takes for a drug to go from conception to market is approximately 15 years. It may sound like a long time, but when all the necessary steps and any set-backs are considered it’s a marvel drugs get made at all. The overall process is sub-divided into a few distinct parts, which in turn have many steps of their own. There is the initial discovery and development, preclinical development, and the clinical trial phase. The key questions to answer in this overall process are- first and foremost is the drug safe and second, does it work. The clinical trials are the final step in determining whether the drug is approved by the FDA and made available to the public. That is not to say the research stops after it hits the public, there is the post-marketing research and continued safety trials. The company that makes the drug will never really stop researching it, there is always work being done behind the scenes long after the FDA approval. The main idea behind clinical trials is they are a way to test the drug in humans, specifically those with the disease or condition the drug is indicated to treat.
There are four clinical trial phases- the first three are pre-market and the fourth is conducted when the drug is on the market. The first phase sets out to answer one of the two questions mentioned above- Is the drug safe? To do this, phase one generally enroll healthy individuals who do not have the disease of interest and overall are relatively healthy. This first phase’s goal is to determine if people can take the drug and maintain their health. This is the first introduction of the drug into human subjects. Along the way in the discovery and preclinical stages the drug is exposed to a variety of cell types and animals- mice, rats, and dogs for example. All of these animal studies are carefully looked at and if there are any safety concerns in these species, studies would not be run on human subjects. However it is still a brave individual who signs up for Phase I studies. Of course the individuals are closely monitored; they get regular physical examinations and answer extensive questions about all aspects of their health.
Interestingly this is how Viagra came about- Pfizer was testing a compound to treat angina and the patients enrolled in Phase I were experiencing some unusual side effects-prolonged erections to be exact. In addition to this unanticipated side effect it turns out the drug didn’t really work well on angina. Pfizer stopped the trials aimed at angina and re-started with trials aimed at erectile dysfunction. I was glad to read that this was an accidental discovery since I didn’t understand why with all the diseases out there in need of treatment a company would intentionally go after erectile dysfunction. Can’t say I blame them for going ahead with the new disease indication and making tons of money off of it. Especially if that money helps them make drugs for life-threatening diseases.
Phase two clinical trials introduce the drug to the intended market- people suffering from the disease of interest. Phase II sets out to answer the question does the drug work. This involves using a small group of patients with the disease and treating them in a very controlled manner. Just because the overall question of safety is answered in Phase I does not mean safety is no longer looked into, the direct opposite in fact. Safety is continued to be the major factor in all of these studies. Phase III is an expanded version of Phase II, now that the safety and efficacy have been shown this stage goes into greater detail in terms of the people in the study. A broader range of subjects are looked at- again people with the disease but now the number of people is dramatically increased and the ranges in terms of age and gender, if applicable, for the given disease.
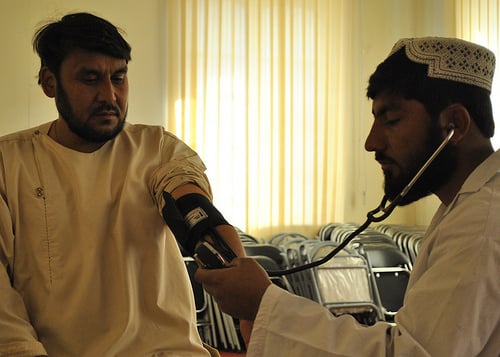
Additional information gained from the trials includes side-effects, which are closely monitored in each phase. In phase I side-effects of healthy patients are examined in correlation to the dosing. In stages II and III, side effects are monitored to gauge the risk to benefit. The risk to benefit varies depending on what the drug is used for- for example the way I think of it is I’d rather have an unplanned pregnancy instead of a fatal blood clot. A few years ago there was a new form of birth control for women, that also treats PMDD (Premenstrual Dysphoric Disorder) but after being out on the market for a while the drug was found to cause fatal blood clots in several young women. For a pharmaceutical that functions as birth control, or the above example of Viagra, the risk to benefit ratio needs to be pretty low since it is not a life-saving drug. Take cancer- it devastates the human body but the current standards of treatment, chemotherapy and radiation, are unfortunately not so gentle on the body either. Side effects for a cancer drug are allowed to be somewhat harsher than a drug set out to treat non-life-threatening conditions because the benefit of ridding the body of cancer outweighs the risk of side-effects caused by the treatment. In addition to looking at and monitoring side-effects, dosing information is looked into in these trials as well. It is first looked at in the healthy patients in Phase I, then continued to be worked on in Phase II where frequency of dosing is examined. The general dosage to start in Phase I is supplied by the pre-clinical trials in which other species are the subjects.
The ideal type of study to run is a randomized, controlled, double-blind study. A controlled study has both an experimental group, which receives the new medicine or treatment, and the control group which does not receive the drug or therapy. The trial is randomized by randomly putting patients into the either the experimental or control group. In a non-random trial the groups would be arranged based on specific criteria, for example purposely putting an equal number of females in both the control and experimental arm of the study. The double-blind portion means both the participants and the administrators do not know which group is which. A single-blind study, typically, is when the administrators know which group a given patient is in, while in an open study both the patients and the administrators know who is in the different groups. Double-blind is the ideal since bias, even unintentional, can occur if the researchers know which patient is in which group. Likewise it is better for the patients to remain in the dark about which group they are in, as to not bias their responses to health related questions. In Phase III the standard of care is used as the control whenever possible. This means if there is a current treatment for the disease, the new drug is directly compared to it to see if there is improvement upon it. Unfortunately in diseases where there is not currently a treatment, the control subjects get a placebo. Studies are done with the utmost concern for the health and safety of the people enrolled in them- if a non-treatment group, the control group, is not safe and ethical then it is not used.
On a bit of a side note, cancer clinical trials are in a league of their own. These trials raise important questions about trial design and the use of placebos. The standard treatment for cancer is chemotherapy or radiation and currently there are many small molecule inhibitors being researched which are administered in the pill form. This brings up my question of how a study is blinded when the standard therapy is administered intravenously and the new treatment is oral. I came across an informative and interesting article on the National Cancer Institute’s website ‘Design Dilemmas- The Debate over Using Placebos in Cancer Clinical Trials’ from May 2011. Here is the link. It answered my question, one way is to give chemotherapy in addition to the new, orally administered drug and in the case of the control give chemotherapy and an oral placebo. This is an ideal situation because the placebo patients are still getting the standard treatment. But this only works if the researchers are interested in giving the drug in combination with chemo and/or can get the answers they need by giving the combination. In addition to answering my question this article gave a lot of information explaining the idea of placebo use in cancer.
When all of the necessary trials for phase I, II, and III are run- each candidate is different and may have several variations of trials run under the umbrella of these three categories-the NDA is filed. The New Drug Application is submitted to the FDA containing all of the preclinical and clinical data. The application is reviewed, often times more information and possibly more studies are requested from the FDA and best case scenario- the drug gets approval. The fourth stage of trials involves the company continuing to look into the fundamentals- is the drug safe, is it working, and what is the risk to benefit scenario regarding the side-effects. This monitoring allows for an even broader group of patients than the Phase III and often times this is when dangerous side-effects are fully elucidated. This can lead to the drug being pulled from the market. Phases I through III are as thorough as they can be, but sometimes risks are not fully illustrated until it is taken by the general public.
Personally, this would make me apprehensive of taking a new medicine that has been on the market for less than 5 years. One recent example was Zelnorm, which treated (IBS) - Irritable Bowel Syndrome-it was pulled off the market after one year due to heart attack and stroke in patients taking the drug. The birth control I wrote about earlier, YAZ, has yet to be pulled but seems to be on its way due to blood clots, heart attacks, and gallbladder disease. (Currently the FDA has made Bayer beef up the warnings on the packaging in order to emphasize the risks associated with taking it, this black box warning is the most serious warning the FDA can issue). Unfortunately there is no way to know how people in general will react to a drug, until it is on the market and fully accessible to a wide variety of individuals.
Sources:
http://www.alzforum.org/drg/tut/tutorial.asp
http://consumerjusticegroup.com/drug-recalls/recalltimeline/