How carbon in diamonds is returned to nature and become part of natural Carbon C
How carbon in diamonds is returned to nature and become part of natural Carbon Cycle?
Carbon is the backbone of life on Earth. We are made of carbon, we eat carbon, and our civilizations. And transformation of carbon from one farm to another is a thing which keeps life going on, and which is generally referred to as Carbon Cycle. My question is that is the carbon in diamonds, is also part of Carbon Cycle? and if yes bywhich mehods.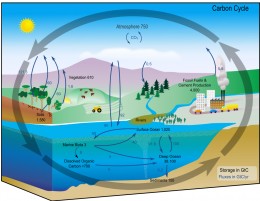
I suppose you mean that diamonds, being relatively indestructible by ordinary means, will not be 'recycled' into other forms of carbon?
I suppose further that the basic answer is 'no.' You can burn diamonds if you have a hot enough flame--1520 F is mentioned on one website. But under most probable scenarios, they will be around essentially forever--they won't decay or degrade, and if broken or crushed you just get smaller diamonds. So diamonds aren't being 'recycled' in any meaningful way.
But diamonds won't 'take over' the carbon cycle for 2 reasons:
1) they form very slowly--one estimate says diamonds in general may be 1/4 of the age of the Earth itself. And they form deep in the Earth's mantle, so their carbon was 'already withdrawn' from the carbon cycle.
2) The amount of carbon in diamonds is a tiny fraction of the total carbon in the world. The total diamond production of South Africa, the world's leading producer, is about 10 million carats. In kilograms, that's about 5,000, if I haven't dropped a decimal somewhere. (A carat is .2 grams.) By contrast, world emissions of carbon due to fossil fuel combustion reached 10 billion tonnes in 2010. In kilograms, that would be 10 followed by 12 'zeros.' So the ratio of diamonds to carbon emissions would be about 2 billion to 1--that's roughly you (or I) compared to the total populations of India, the US, Indonesia and Brazil. And human emissions are much smaller than the total carbon in the biosphere, let alone all the geologically-stored carbon.
So more carbon is being (re-)introduced into the cycle all the time by human activities. There's no danger of running out--though there is a clear danger that we will continued to disrupt our climate with all the CO2, which is a greenhouse gas.Diamonds are actually a crystallised form of the element carbon which, when exposed to extreme temperatures and pressure, forms natural diamonds deep beneath the earth’s crust.
The formation of diamonds begins around 90 miles into the earth, where temperatures reach in the region of 1200 degrees. Diamonds can also form as a result of other natural events.
Source: Quantum International Diamonds
Related Discussions
- 107
Many Many Little Tiny People (and Cows) are Causing Global Warming.
by Kathryn L Hill 4 years ago
The earth is so huge. And people are so small. Are we really contributing to the build-up of C02 in the upper atmosphere(s) by:1. Burning fossil fuels, 2. Cutting down forests 3. Farming livestock.https://news.climate.columbia.edu/2021/ … l-warming/"Fossil fuels produce large quantities...
- 32
Do you consider CO2 as an air pollutant?
by Ralph Schwartz 8 years ago
Do you consider CO2 as an air pollutant?According to the World Health Organization (WHO), the U.S. is among the cleanest nations on the planet, with the cleanest air in the world, significantly cleaner in fact than the air in Germany, Italy, Switzerland, the UK, Japan, Austria and...
- 10
What is the rarest color for natural diamonds?
by Beth Perry 11 years ago
What is the rarest color for natural diamonds?Yellow, blue, pink, white, something else? I have often wondered.
- 2
im 19 an in 10th grade i went natural but in 12th grade returned to the perm. a
by understanding jes 14 years ago
im 19 an in 10th grade i went natural but in 12th grade returned to the perm. an decided to cut...my hair in to a short style. now i've decided to return to natural but i do not know where to start or how to start with out looking a mess. where do u suggest i start?






