Here's one for physical science and chemistry geeks..
Here's one for physical science and chemistry geeks..
I understand WHAT surfactants are; what they do. What I do not know, and would like to understand, is HOW they do what they do.
How do they break the surface tension of water; break that bond, without turning the water into a different element by the mixing of the chemical agent with the water?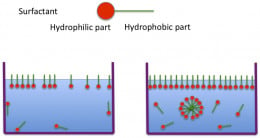
A DIALOGUE WITH SARAH, AGED 3: IN WHICH IT IS SHOWN THAT IF YOUR DAD IS A CHEMISTRY PROFESSOR, ASKING “WHY” CAN BE DANGEROUS
SARAH: Daddy, were you in the shower?
DAD: Yes, I was in the shower.
SARAH: Why?
DAD: I was dirty. The shower gets me clean.
SARAH: Why?
DAD: Why does the shower get me clean?
SARAH: Yes.
DAD: Because the water washes the dirt away when I use soap.
SARAH: Why?
DAD: Why do I use soap?
SARAH: Yes.
DAD: Because the soap grabs the dirt and lets the water wash it off.
SARAH: Why?
DAD: Why does the soap grab the dirt?
SARAH: Yes.
DAD: Because soap is a surfactant.
SARAH: Why?
DAD: Why is soap a surfactant?
SARAH: Yes.
DAD: That is an EXCELLENT question. Soap is a surfactant because it forms water-soluble micelles that trap the otherwise insoluble dirt and oil particles.
SARAH: Why?
DAD: Why does soap form micelles?
SARAH: Yes.
DAD: Soap molecules are long chains with a polar, hydrophilic head and a non-polar, hydrophobic tail. Can you say ‘hydrophilic’?
SARAH: Aidrofawwic
DAD: And can you say ‘hydrophobic’?
SARAH: Aidrofawwic
DAD: Excellent! The word ‘hydrophobic’ means that it avoids water.
SARAH: Why?
DAD: Why does it mean that?
SARAH: Yes.
DAD: It’s Greek! ‘Hydro’ means water and ‘phobic’ means ‘fear of’. ‘Phobos’ is fear. So ‘hydrophobic’ means ‘afraid of water’.
SARAH: Like a monster?
DAD: You mean, like being afraid of a monster?
SARAH: Yes.
DAD: A scary monster, sure. If you were afraid of a monster, a Greek person would say you were gorgophobic.(pause)
SARAH: (rolls her eyes) I thought we were talking about soap.
DAD: We are talking about soap.(longish pause)
SARAH: Why?
DAD: Why do the molecules have a hydrophilic head and a hydrophobic tail?
SARAH: Yes.
DAD: Because the C-O bonds in the head are highly polar, and the C-H bonds in the tail are effectively non-polar.
SARAH: Why?
DAD: Because while carbon and hydrogen have almost the same electronegativity, oxygen is far more electronegative, thereby polarizing the C-O bonds.
SARAH: Why?
DAD: Why is oxygen more electronegative than carbon and hydrogen?
SARAH: Yes.
DAD: That’s complicated. There are different answers to that question, depending on the Pauling or Mulliken electronegativity scales. But it really all comes down to effective nuclear charge. The electrons in an oxygen atom have a lower energy than a carbon atom, and electrons shared between them are held more tightly to the oxygen Cool, huh?(pause)
SARAH: I don’t get it.
DAD: That’s OK. Neither do most of my students.That's hilarious. But thanks for the explanation. I mostly get it! ;-)
Related Discussions
- 2
Is an applied chemitry student eligible for teaching in college in physical chem
by humen gogoi 15 years ago
Is an applied chemitry student eligible for teaching in college in physical chemistry?whether an spplied chemistry(medicinal chemistry specialization) cant teach in any branch of chemistry e.g organic inorganic physical in college?
- 17
Why are some of us attracted to someone who is unavailable ?
by Twila Nelson 14 years ago
Why are many of us attracted into relationships with someone who is unavailable ? Why when we find out the person is not available do we stay in the relationship(all excuses and rationalizations set aside) ?
- 75
Why would anyone in their right mind want Sarah Palin as President?
by tHErEDpILL 14 years ago
These supporters of Sarah Palin must know something I don't know, if so please enlighten me. Otherwise all I have seen from this women is incompetence. In my opinion, this women might be the most unqualified presidential hopeful in the history of the United States of America. We...
- 160
Sarah Palin and the Mainstream Media.
by BJC 16 years ago
Why are some people afraid of Sarah Palin?? For sure she doesn't flow with the media and is definitly going upstream. Could be that some folks are concerned because she has the gonads to speak truth and the far left are aware that she could create some problems? Lets face it, when...
- 115
Why do you like Sarah Palin or Why don't you like Sarah Palin
by woolman60 16 years ago
Just be honest, do you really think she could run America?
- 5
Why is it so hard for a step child to accept you?
by vshining 16 years ago
Why is it so hard for a step child to accept you?







